Background
TAVR Alternative Access and its problems
Needs Statement
There is a clinical need for a way to better adapt the Edwards Lifesciences introducer sheath to alternative access TAVR procedures.
Transcatheter Aortic Valve Replacement (TAVR)
Surgery to replace aortic heart valve with a prosthetic using a catheter system.

TAVR is a minimally invasive surgical procedure that is performed in cath labs in order to replace diseased aortic heart valves with a prosthetic. The prosthetic is a collapsible valve that is deployed using an inflatable surgical balloon. The prosthetic valve is delivered to the heart using a system of guided tubes known as catheters, while the patient is under an anesthetic. The guidance of the system and prosthetic is confirmed using various imaging techniques during the procedure. The catheter system is able to enter safely through small incisions with the help of what is known as an introducer sheath. Generally the catheter enters the femoral artery and is guided upwards into the heart; this is known as transfemoral access. However, in rare instances, TAVR may be performed by entering through alternative arteries, which is known as alternative access.
Patients
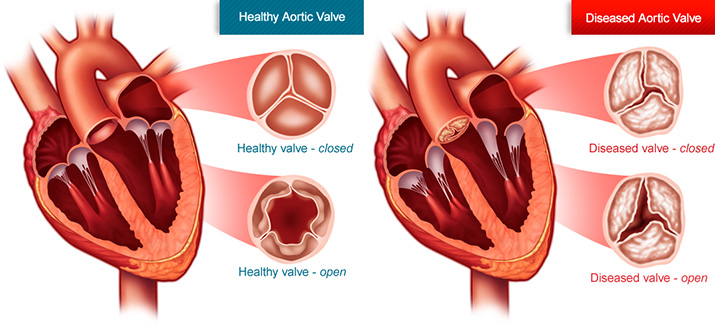
 Heart disease remains to be the leading cause of death in the US. One cause of heart disease is aortic stenosis. Aortic stenosis is a condition in which the aortic heart valve becomes severely calcified over time. This irreversible calcification causes the valve to become stiff and prevent it from fully opening and closing. The result of this reduced blood flow out of the heart, as well as severe levels of blood leaking back into the heart. Both of these cause the heart to overwork in order to maintain proper circulation. This causes the heart to become weaker over time due to the extra strain, ultimately leading to heart failure. About 5% of the population at the age of 65 have aortic stenosis and the prevalence increases further with age. More than 2.5 million people older than 75 years have aortic stenosis in the United states.
Heart disease remains to be the leading cause of death in the US. One cause of heart disease is aortic stenosis. Aortic stenosis is a condition in which the aortic heart valve becomes severely calcified over time. This irreversible calcification causes the valve to become stiff and prevent it from fully opening and closing. The result of this reduced blood flow out of the heart, as well as severe levels of blood leaking back into the heart. Both of these cause the heart to overwork in order to maintain proper circulation. This causes the heart to become weaker over time due to the extra strain, ultimately leading to heart failure. About 5% of the population at the age of 65 have aortic stenosis and the prevalence increases further with age. More than 2.5 million people older than 75 years have aortic stenosis in the United states.
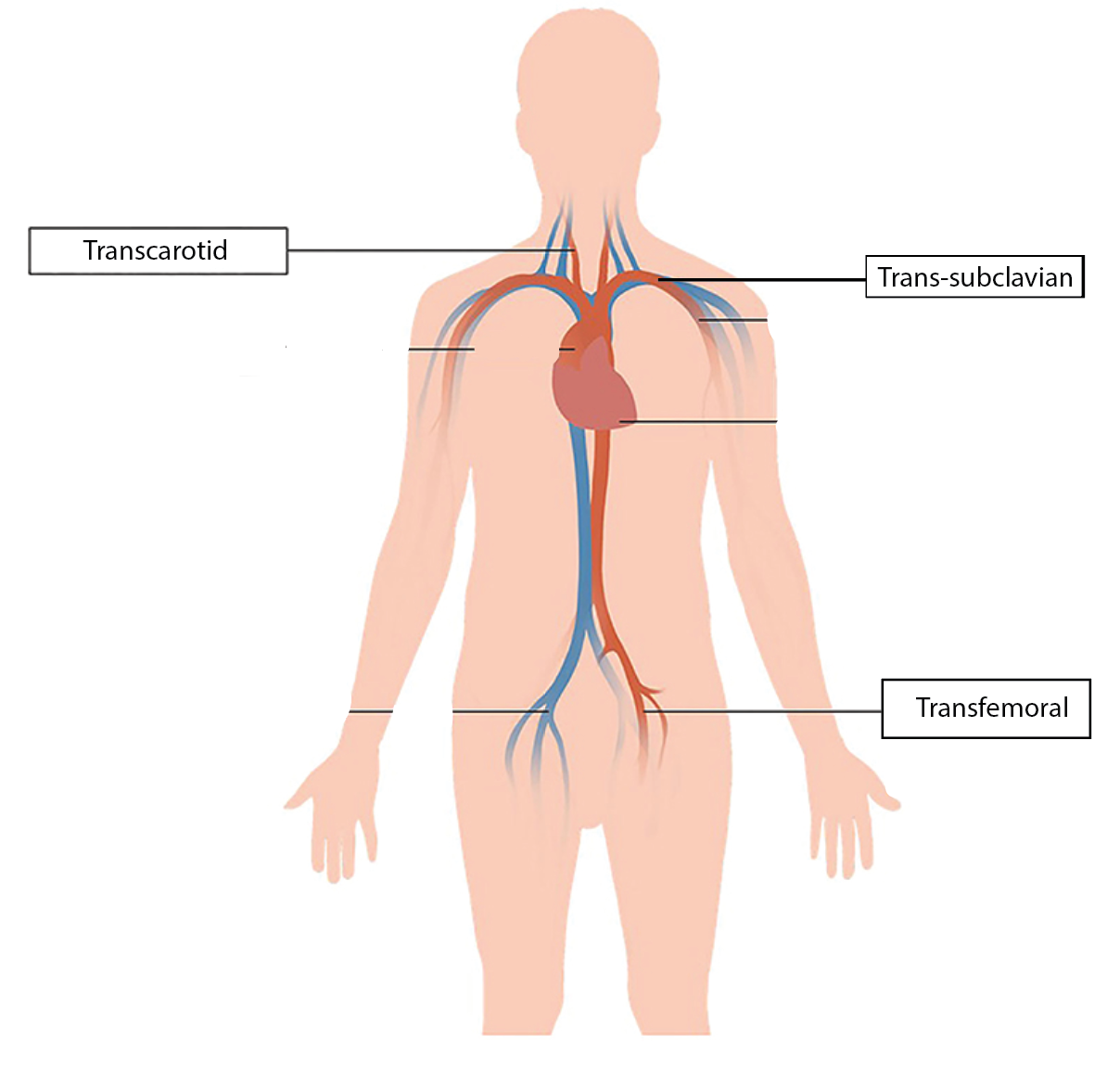
 TAVR is the standard of care for treating aortic stenosis, with over 90,000 cases per year. However, about 10% of these patients are unable to use the common Transfemoral access that the TAVR systems were designed for. This inability to use transfemoral access is because some patients have femoral arteries that have too many bends or too much calcium build up, which makes this method of access potentially fatal. In such cases physicians will use alternative access, by entering through alternative methods. The most common routes of entry for these 9,000 annual patients are the carotid artery (known as transcarotid access) and the subclavian artery (known as trans-subclavian access). However, because the TAVR systems were designed specifically for transfemoral access, physicians have difficulty performing alternative access TAVR, which increases the risk for procedural complications.
TAVR is the standard of care for treating aortic stenosis, with over 90,000 cases per year. However, about 10% of these patients are unable to use the common Transfemoral access that the TAVR systems were designed for. This inability to use transfemoral access is because some patients have femoral arteries that have too many bends or too much calcium build up, which makes this method of access potentially fatal. In such cases physicians will use alternative access, by entering through alternative methods. The most common routes of entry for these 9,000 annual patients are the carotid artery (known as transcarotid access) and the subclavian artery (known as trans-subclavian access). However, because the TAVR systems were designed specifically for transfemoral access, physicians have difficulty performing alternative access TAVR, which increases the risk for procedural complications.
Current Alternative Access Problems
The Edwards Introducer eSheath system is specifically designed with a length that would only be appropriate for transfemoral access. Performing TAVR in the alternative access cases using the current Introducer eSheath system is difficult and strenuous on the operating physicians. Currently, physicians do not have any devices to assist with improving alternative access TAVR.
Edwards Introducer eSheath
In the TAVR procedure, the introducer eSheath acts as a tunnel in and out of the body for the physician to be able to insert tools used for the procedure, without damaging the patient’s artery walls. The sheath contains one way seals at its base that will mitigate bleeding, and a specialized tip to allow for smooth entry into the artery to prevent damage. If the clinician were to proceed with an alternative access with the current introducer system there are risks with damaging the vasculature and over penetrating the heart, if no precautions are taken.

Issues Regarding Sheath Length
Sheath length should vary greatly depending on the access route for the procedure. Edwards eSheath platform is currently only available for the transfemoral access route; the length of the Edwards sheath is much longer than the length needed for other alternative access procedures. Should a patient need a transcarotid or trans-subclavian TAVR procedure, there will be a large portion of the sheath outside the body.
| Access Type | Required Length |
|---|---|
| Edwards Sheath (Transfemoral Access) |
37 centimeters |
| Transcarotid | 20 centimeters |
| Transubclavian | 17 centimeters |
Presently, there are only a few ways that hospitals have reduced the risks to patients by devoting one operating room staff to holding and securing the sheath in position. Cath labs, where TAVR is performed, are extremely limited in space, so this method of securing is highly wasteful of valuable resources. Moreover, having a person hold it is entirely unreliable as they may shake, be bumped, or their grip may slip, which may cause severe complications. This method also makes the procedure much more difficult and straining on the operating staff, which can increase the chance of procedural complications.
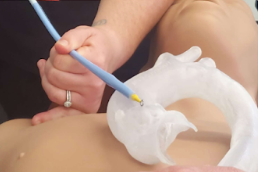 The other way that the procedure has been done is by cutting off the tip of the eSheath. However, this comes at a cost of removing the specialized tip, which is designed to reduce damage to the vasculature. Both current fixes are only patches, and require a long term solution that is consistent and repeatable to reduce the risks to patients as much as possible.
The other way that the procedure has been done is by cutting off the tip of the eSheath. However, this comes at a cost of removing the specialized tip, which is designed to reduce damage to the vasculature. Both current fixes are only patches, and require a long term solution that is consistent and repeatable to reduce the risks to patients as much as possible.
